Generic Abortion Pill Approval Fuels Anti-Abortion Outrage
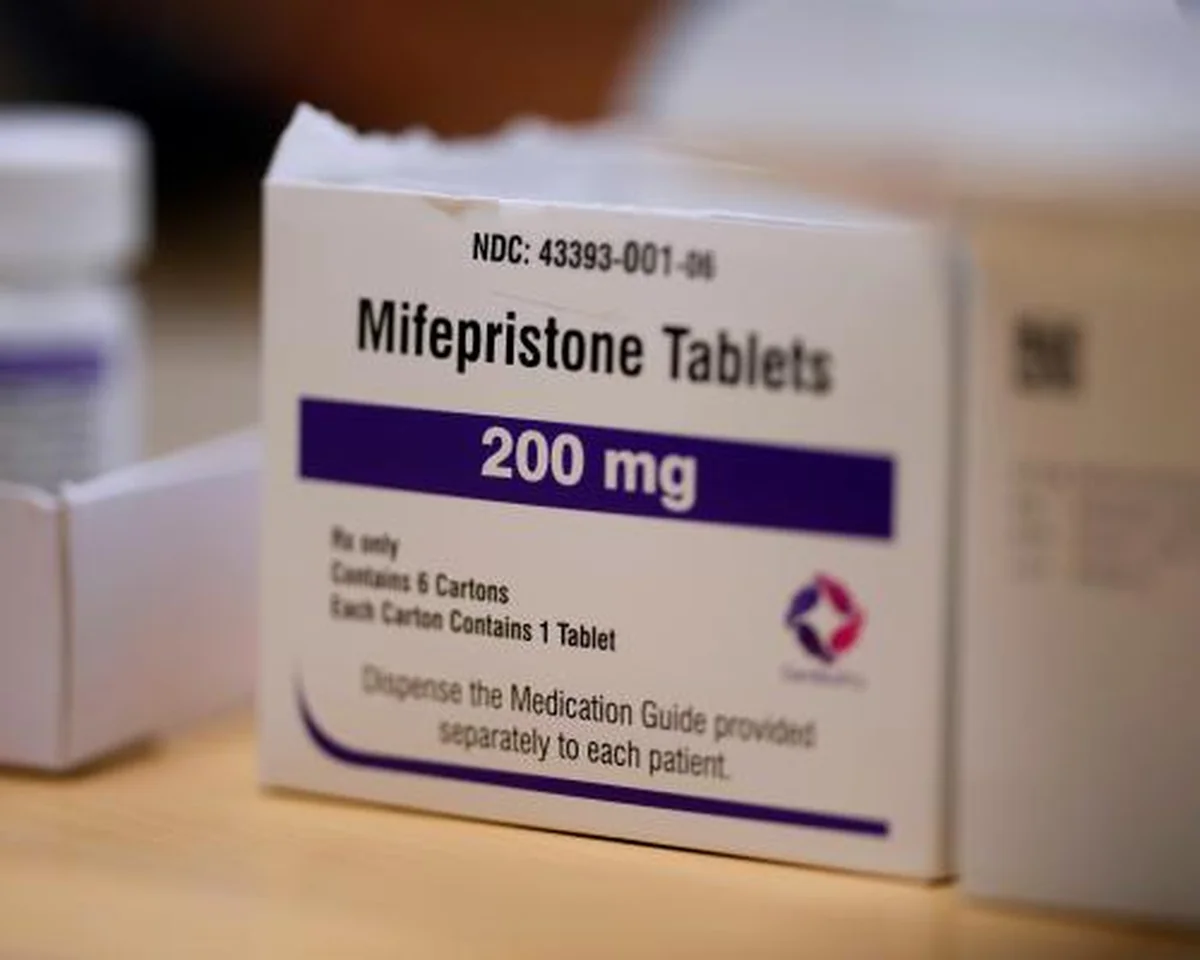
The U.S. Food and Drug Administration (FDA) has ignited a fresh wave of controversy following its quiet authorization for a new generic version of mifepristone, a key component in most medication abortions. This decision, which allows a company named Evita Solutions to manufacture its alternative to the brand-name drug first approved in 2000, has been met with immediate and fierce condemnation from anti-abortion organizations and advocates across the nation.
Context of the Approval
Mifepristone, one of two medications used in combination for medical abortions in the United States, already has a generic counterpart available on the market. However, the FDA’s latest move comes amidst a highly charged political environment. In the three years since the landmark overturning of Roe v. Wade in 2022, anti-abortion proponents have intensified their lobbying efforts, urging the federal government to restrict access to the drug, making this new approval particularly galling to them.
Political Figures React
The announcement prompted swift and scathing criticism. Senator Josh Hawley (R-Missouri), a vocal opponent of abortion access, expressed his dismay on X, stating, “FDA had promised to do a top-to-bottom safety review of the chemical abortion drug, but instead they’ve just greenlighted new versions of it for distribution. I have lost confidence in the leadership at FDA.” This sentiment was echoed by Kristan Hawkins, president of Students for Life of America, a prominent anti-abortion group, who branded the approval “a true failure” and, in a statement, went further to call it “a stain on the Trump presidency.” The timing also follows Health Secretary Robert F. Kennedy Jr.’s recent announcement that his department would initiate a safety review of mifepristone.
Controversial Safety Claims
Central to the renewed offensive against mifepristone is a recent paper published in April by the Ethics and Public Policy Center, a conservative think tank. Anti-abortion activists have vigorously promoted its claims, which allege that nearly 11% of women experience severe complications, such as sepsis, within 45 days of taking the medication. This report was notably cited by Secretary Kennedy in his letter outlining the planned review of the drug’s safety profile.
Experts Debunk Flawed Research
However, the veracity of these claims has been widely questioned by medical experts. The paper from the Ethics and Public Policy Center has neither undergone peer review nor been published in a recognized medical journal. Furthermore, specialists have identified significant methodological flaws within the report. A key example includes its classification of ectopic pregnancies—where an embryo develops outside the uterus—as a serious complication directly linked to mifepristone. Medical consensus confirms that mifepristone does not cause or exacerbate ectopic pregnancies; rather, it is ineffective in treating them and requires different medical intervention.
The FDA’s decision to broaden the availability of generic mifepristone underscores the ongoing, deeply polarized battle over reproductive healthcare access in the United States. While federal regulators appear to be moving forward with expanding options for medication abortion, anti-abortion forces continue to leverage every available avenue, from political pressure to contested research, in their relentless campaign to restrict access to these drugs.
Source: The Guardian